Metformin ER + Low‑Dose Naltrexone


500mg/tablet - 240 tablets (3mo supply).
Free expedited shipping.
No membership fees.
Doctor-led plans, coaching & active community.
Provider tailored plans for real-life needs
Every plan is personalized to support your unique biology and lifestyle. Providers may include medications for:
Appetite awareness
Blood sugar support
Mood & craving support
Digestive and metabolic routine
Energy & focus
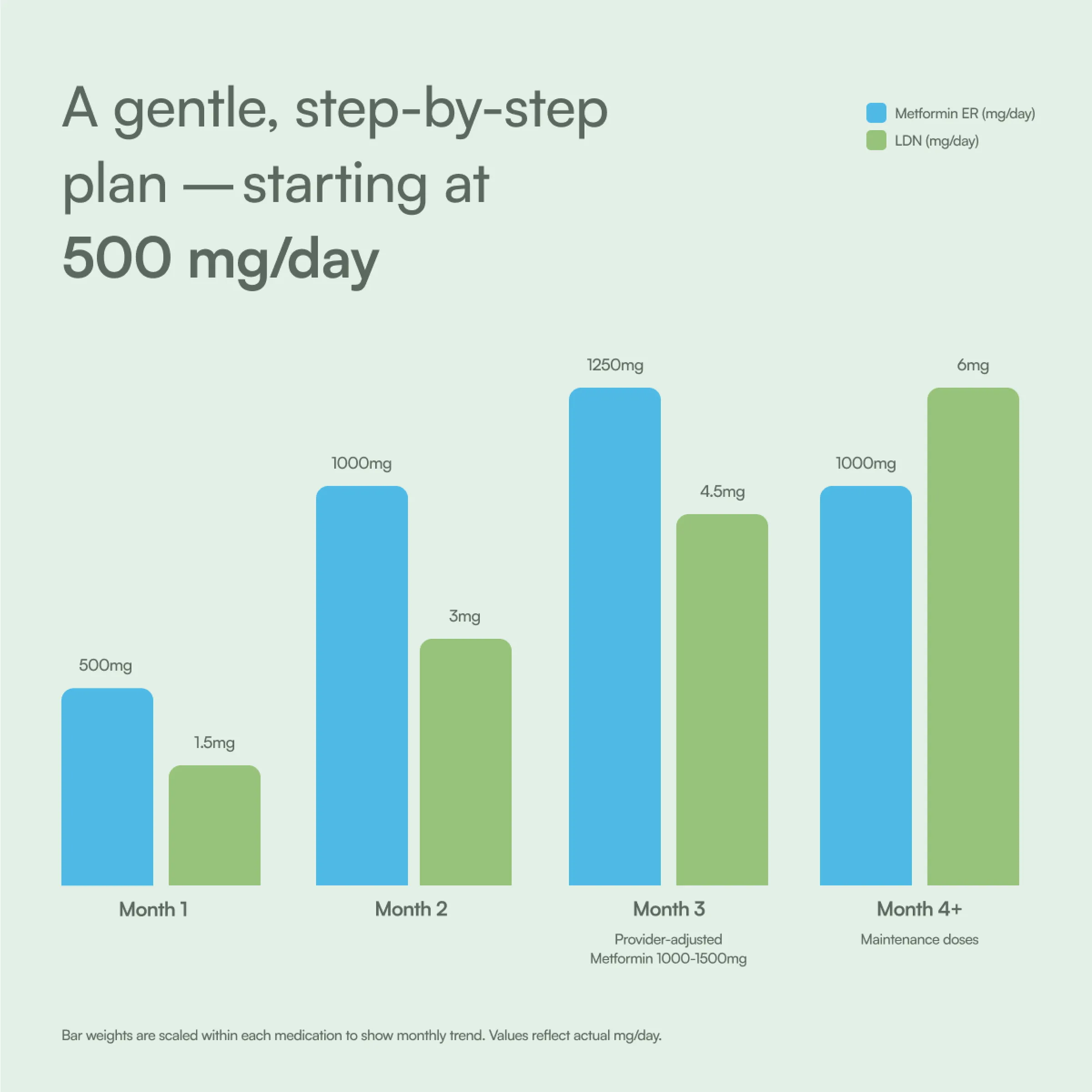
Your Metformin ER + Low‑Dose Naltrexone dosage schedule
Your dosage increases gradually over time to support better tolerability and sustained metabolic results.
Proven results, lab tested
Our medication is delivered from a state licensed pharmacy in our network, right to your door when you need it.
Our pharmacies perform third party testing through FDA and DEA registered labs to run quality control checks for every compounded lot prescribed for Eden patients. They test four key characteristics that are associated with quality and safety.
More from our members
Individual results may vary. Testimonials reflect personal experiences and do not guarantee outcomes.
Learn more about Metformin ER + Low‑Dose Naltrexone
This combination is designed to support appetite awareness, cravings management, and balanced energy as part of a provider-guided plan. Metformin ER (extended-release) supports glucose balance and metabolic health, while low-dose naltrexone (LDN) may help regulate appetite and cravings when prescribed off-label.
No. Metformin is FDA-approved for type 2 diabetes but not for weight loss. LDN is not FDA-approved for weight loss or obesity treatment. Both may be prescribed off-label by a licensed provider based on clinical judgment and individual health goals.
- Metformin ER: Reduces sugar production in the liver, improves how the body uses insulin, and helps stabilize energy levels. Some studies suggest it may indirectly support appetite regulation in certain individuals.
- LDN: Works on endorphin and opioid receptor pathways, which may influence appetite regulation, cravings, and inflammation.
Together, these may complement one another under provider supervision to support sustainable lifestyle changes.
Some members report:
- More stable energy from meals
- Greater awareness of appetite cues
- Reduced cravings or emotional eating triggers
- Improved sense of balance when transitioning off dieting or other therapies
Results vary and depend on health status, adherence, and provider guidance.
- Metformin ER: The most common side effects are digestive issues such as nausea, diarrhea, or stomach upset. Long-term use may lower vitamin B12 levels.
- LDN: May cause vivid dreams, nausea, or mild headaches in some individuals. These effects are typically dose-dependent and may improve with adjustment.
Metformin can lower vitamin B12 over time, which is why providers often recommend monitoring or supplementing. LDN may interfere with opioid-based medications and is not suitable for people using opioid pain treatments. Always share your full medication and supplement list with your provider before starting.
Neither Metformin nor LDN are FDA-approved for weight loss. Some studies suggest Metformin may support modest weight reduction in certain populations when used off-label. LDN’s role in weight management is still under study and not clinically established. Any benefits depend on individual health factors, adherence, and provider supervision.
This combination may not be suitable for individuals who are:
- Pregnant or breastfeeding
- Living with severe kidney or liver disease
- Experiencing recurrent vitamin B12 deficiency
- Currently using opioid medications or with opioid dependence
- Managing uncontrolled heart disease or hypertension
Your provider will carefully review your history before prescribing.
Only available if prescribed after an online consultation with a healthcare provider. Benefits outlined are based on third-party studies. Plans are offered as a subscription service which can be canceled at any time. Actual product packaging may appear differently than shown. The FDA does not review or approve any compounded medications for safety or effectiveness. These treatments are individualized and do not guarantee specific results. Provider-determined, off-label use may apply.
The statements on this page have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure or prevent any disease.
Important safety information- The New England Journal of Medicine: Reduction in the Incidence of Type 2 Diabetes with Lifestyle Intervention or Metformin
- American College of Physicians: Annals of Internal Medicine: Long-Term Weight Loss With Metformin or Lifestyle Intervention in the Diabetes Prevention Program Outcomes Study
- Oxford Academic: Human Reproduction: Metformin reduces pregnancy complications without affecting androgen levels in pregnant polycystic ovary syndrome women: results of a randomized study
- The Lancet: Effect of naltrexone plus bupropion on weight loss in overweight and obese adults (COR-I): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial
- Obesity: A Research Journal: A Randomized, Phase 3 Trial of Naltrexone SR/Bupropion SR on Weight and Obesity-related Risk Factors (COR-II)
- Obesity: A Research Journal: Bupropion SR enhances weight loss: a 48-week double-blind, placebo- controlled trial
- Obesity: A Research Journal: Bupropion for weight loss: an investigation of efficacy and tolerability in overweight and obese women
- American Heart Association: Stroke: Acarbose slows progression of intima-media thickness of the carotid arteries in subjects with impaired glucose tolerance
- Journal of the American Medical Association: Weight Control and Risk Factor Reduction in Obese Subjects Treated for 2 Years With Orlistat
- American College of Physicians: Annals of Internal Medicine: Long-term Weight Loss with Metformin or Lifestyle Intervention in the Diabetes Prevention Program Outcomes Study
- Obesity: A Research Journal: Weight Loss With Naltrexone SR/Bupropion SR Combination Therapy as an adjunct to Behavior Modification: The COR-BMOD Trial
- Mayo Clinic: Pyridoxine (oral route, injection route)
- BMJ Journals: Openheart: Myo-inositol for insulin resistance, metabolic syndrome, polycystic ovary syndrome and gestational diabetes
- Mayo Clinic: Acarbose (oral route)
- StatPearls: Orlistat
- National Institutes of Health: Carnitine
















.webp)
.webp)
.webp)
.webp)